Product Compliance
filing/registration/consulting/training

The cosmetic formula sheet is a core document in the product registration and filing process. Its accuracy and compliance directly impact a product's ability to pass technical review. To help companies accurately submit formula information and avoid common errors, this article systematically outlines the requirements and precautions for completing formula sheets, based on relevant regulations such as the "Regulations on the Supervision and Administration of Cosmetics," the "Administrative Measures for the Registration and Filing of Cosmetics," and the "Technical Specifications for Cosmetic Safety."
Formula form filling
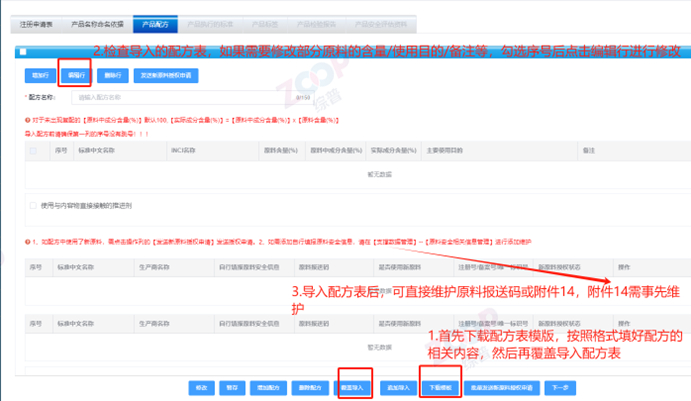
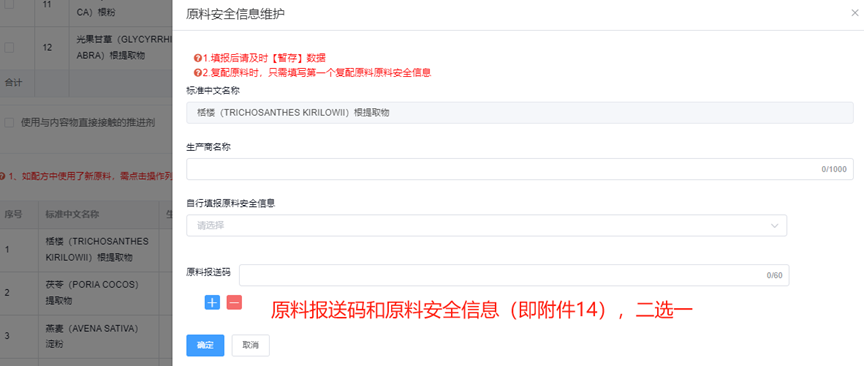
Precautions
1. Ingredient Names
Formula ingredient names must use the standard Chinese names, INCI names, or English names listed in the Ingredient Catalog. If a new cosmetic ingredient currently under safety monitoring is used in a formula, the registered or filed new ingredient name should be used. For ingredients used as colorants, the Colorant Index Number (CI number) listed in the Cosmetic Safety Technical Specifications should be entered in the Ingredient Name column, unless a CI number is unavailable. If a lake is used as a colorant, "(Lake)" should be added after the colorant name. If a nano-ingredient is used, "(Nano-Grade)" should be added after the ingredient name.
2. Raw Material Percentage
Formula ingredients should be listed in descending order of percentage, expressed as a percentage by mass, with the total raw material content totaling 100%. If the formula contains a propellant that comes into direct contact with the product contents, the propellant composition and content should be reported separately, with the total propellant content totaling 100%. The fill ratio of propellant to material should also be indicated.
3. Purpose of Ingredients
Registrants of cosmetics should label the primary purpose of the ingredient based on its actual function in the product. Cosmetics claiming hair dyeing, perming, freckle removal and whitening, sun protection, hair loss prevention, acne treatment, anti-wrinkle, anti-dandruff, or deodorization, as well as cosmetics claiming novel functions (except for those intended for special populations), should list the corresponding active ingredient in the Purpose column of the formula. If the active ingredient requiring labeling is not a single component, the specific active ingredient should be clearly stated in the Purpose column.
4. Recipe Table Notes
Any further information regarding the ingredients listed in the recipe should be provided in the Recipe Table Notes column.
For example:
● If hydrocarbons (except single components) derived from petroleum or coal tar are used as raw materials, the Chemical Abstracts Index (CAS number) of the raw materials shall be indicated;
● If denatured ethanol is used as raw material, the name and amount of the denaturant shall be stated;
● If category raw materials are used, the specific names of the raw materials shall be stated;
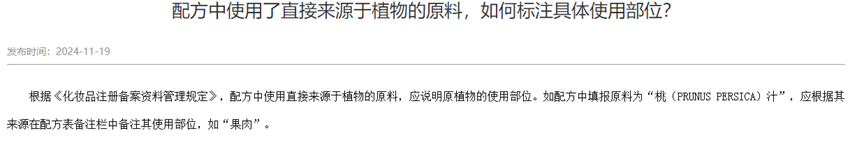
● Use raw materials that are directly derived from plants, and indicate the specific parts of the original plant to be used according to the instructions in the Raw Material Catalog;
● If the product formula only lists "fragrance" as an ingredient, or if the product label identifies the specific fragrance components in the fragrance, or if the original packaging label of an imported product indicates that it contains specific fragrance components, an explanation should be provided in the remarks column of the formula.
● If the ingredients in the formula have the same name but are filled in separately, briefly indicate the reason for filling them in separately in the remarks column of the formula table, such as the ingredients having different molecular weights or different trade names.
● If the colorant used is a lake, the type of lake used should be stated;
● If stickers or films are used as carrier materials, the material composition of the main carrier materials should be indicated in the remarks column, and information such as their source, preparation process, and quality control indicators should be provided;
● If animal organ tissues and blood product extracts are used as raw materials in the product formula, their source, composition and preparation process shall be provided, and relevant documents permitting their use in the country of raw material production shall be provided.
Case



FAQs



Completing a cosmetic formula sheet is a rigorous technical task, involving multiple aspects, including regulations, raw materials, and processes. Companies should strengthen their understanding of relevant regulations and establish robust internal review mechanisms for raw materials and formulas to ensure the authenticity, compliance, and completeness of formula information, laying a solid foundation for a successful product launch.
If you encounter specific questions during the submission process, please contact us for accurate guidance.